|
Research Ideas and Outcomes :
Research Idea
|
|
Corresponding author: Marco Cirillo (marcocir8@gmail.com)
Received: 22 Dec 2020 | Published: 30 Dec 2020
© 2020 Marco Cirillo
This is an open access article distributed under the terms of the Creative Commons Attribution License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation:
Cirillo M (2020) Ring-first Mitral Valve Repair. Research Ideas and Outcomes 6: e62369. https://doi.org/10.3897/rio.6.e62369
|
|
Abstract
Mitral valve repair is one of the most frequent interventions in cardiac surgery. It involves eliminating the dysfunctional part(s) of the mitral valve and reconstructing, using the residual tissue or with the addition of prosthetic components, a properly functioning valve, without residual stenosis or regurgitation. A fundamental component of mitral repair is the implantation of a ring (annuloplasty) which reconstitutes the normal, saddle-shaped geometry of the valve. Such ring is usually implanted at the end of the surgical reconstruction regardless of the repair techniques. The implantation of the ring can however change the final anatomy of the valve in an unexpected way and therefore force new corrective surgical actions. We therefore propose a research project that plans the execution of annuloplasty as the first surgical step and then the correction of the valvular disease affecting the leaflets and chordae. The sizing of the ring is always performed on parts of the valve that are usually not changed during the reconstructive surgery, therefore it is possible to decide its size before surgically correcting the valve. In this way we could act on the leaflets and chordae in the definitive geometrical arrangement of the mitral valve.
Keywords
Mitral valve repair, cardiac surgery, annuloplasty, chordal flail
Overview and background
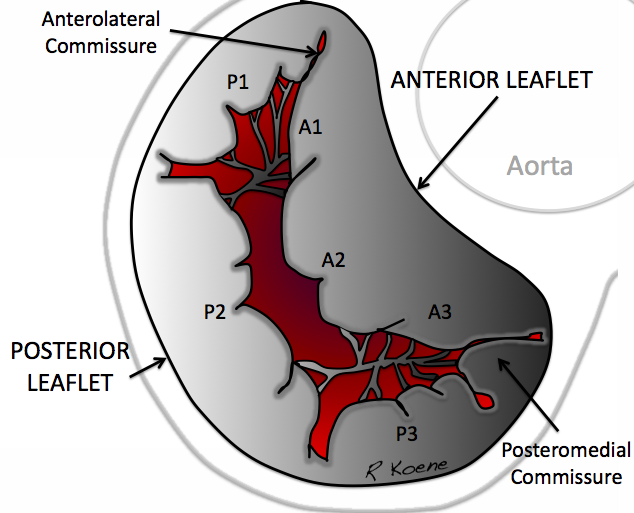
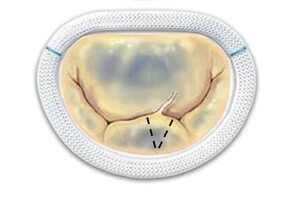
The mitral valve is the atrioventricular valve of the left heart and allows the passage of blood from the left atrium into the left ventricle, preventing its regurgitation in the systolic phase of the cardiac cycle (Fig.
Normal anatomy of the left heart atrioventricular mitral valve (from https://commons.wikimedia.org/wiki/File:Mitral_Valve_RK.png, licensed CC0).
Since Carpentier's seminal works of the 1970s and 1980s and his pathophysiological classification of mitral disease [
This research project is based on the implantation of the prosthetic ring before mitral repair (ring-first repair), to be able to repair the valve already in its definitive geometric arrangement.
Objectives and Project layout
The main objectives of implanting the ring as the first surgical step are:
a) judge the necessary corrections in the final valve geometry;
b) prevent the repaired valve tissue from being abundant (still prolapsing) or scarce (restricted) after the placement of the ring at the end of the surgical correction;
c) avoid having to add extra sutures on leaflets or valve clefts after implantation of the ring, increasing the overall stiffness of the valve.
This procedure is compatible with cases where the ring is implanted prior to valve repair anyway (artificial chordae) and can be limited in cases of anular plication or small mitral valve (rare in valvular regurgitation disease).
The research project has the following key phases:
1) measurement and implantation of the suitable prosthetic ring before repair (ring-first);
2) completion of mitral repair according to the choice of each surgeon.
The size of the ring is always based on the anterior leaflet [
Implementation
The project can easily be developed within a prospective multicenter study with voluntary participation and may lead to a peer-reviewed publication in a surgical journal.
Impact
This project slightly alters the habits of individual operating surgeons adding the great advantage of being able to judge mitral valve repair in the definitive geometric shape of the valve itself, with a more reliable overview (Fig.
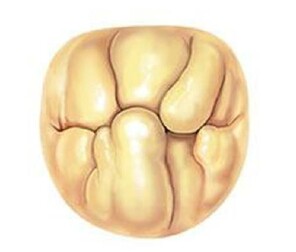
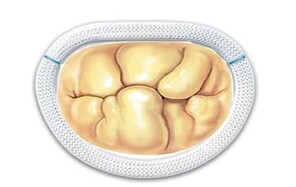
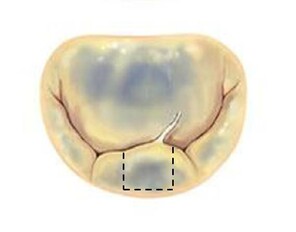
The two most frequent types of mitral pathology: Barlow's disease (a and b) and fibroelastic deficiency (c and d). The pathological pictures are shown and the advantage of implanting the prosthetic ring before performing the repair of the valve leaflets is illustrated.
b: By implanting the ring as a first step, the valve is arranged in the correct elliptical shape, the leaflets are spatially redistributed, then the judgment on any other necessary surgical act is taken in the final shape of the valve.
c: In fibroelastic deficiency the mitral annulus is dilated by chronic regurgitation, widening the valvular area and this may induce a quite large (quadrangular, dashed lines) resection;
d: By implanting the ring as the first step, the valve area is reduced and assumes a more correct geometry, therefore the resection can be less extensive (triangular, dashed lines).
Furthermore, a valve test is performed during each mitral valve repair surgery by filling the ventricle with saline solution: this inflates the valve and allows to better evaluate the type of lesion. By implanting the ring first, the valve area is reduced and the test can therefore be more reliable. Finally, it is very important to avoid adding sutures that weld parts of the leaflets or scallops together [
References
-
The most influential papers in mitral valve surgery; a bibliometric analysis.J Cardiothorac Surg15https://doi.org/10.1186/s13019-020-01214-y
-
Sizing for Mitral Annuloplasty: Where Does Science Stop and Voodoo Begin?Ann Thorac Surg3013; 95:1475‑1483. https://doi.org/10.1016/j.athoracsur.2012.10.023
-
Cardiac valve surgery--the French correction.J Thorac Cardiovasc Surg86(3):323‑337. https://doi.org/10.1016/S0022-5223(19)39144-5
-
Mitral Valve Repair.Operative Techniques in Thoracic and Cardiovascular Surgery3(2):109‑125. https://doi.org/10.1016/S1522-2942(07)70080-6
-
Commissural Closure for Repair of Mitral Commissural Prolapse.Ann Thorac Surg(oracsur.2004.04.051)1135‑1136. https://doi.org/10.1016/j.athoracsur.2004.04.051
-
Mitral valve repair over five decades.Ann Cardiothorac Surg4(4):322‑334. https://doi.org/10.3978/j.issn.2225-319X.2015.01.07
-
Mitral valve annuloplasty: a quantitative clinical and mechanical comparison of different annuloplasty devices.Ann Biomed Eng40(3):750‑761. https://doi.org/10.1007/s10439-011-0442-y
-
Mitral Valve Repair: The French Correction Versus the American Correction.Surg Clin North Am97(4):867‑888. https://doi.org/10.1016/j.suc.2017.03.009
-
Techniques for Repairing Posterior Leaflet Prolapse of the Mitral Valve.Journal of Thoracic and Cardiovascular Surgery - Operative Techniques11(001). https://doi.org/10.1053/j.optechstcvs.2011.11.001